ماء فلوريدي
الماء الفلوريدي بالإنجليزية Fluoridated water أو الفلورة Fluoridation هو إضافة أملاح الفلوريد إلى مياه الشرب بقصد وقاية الاسنان من التسوس. ويؤثر الماء المضاف إليه أحد أملاح الفلوريد مباشرة على مينا الأسنان، ويعدل من مستوى الفلوريد في اللعاب ويحفظ الأسنان بذلك من فقدان مينا الأسنان للأملاح المعدنية.
وقد أدخلت بلاد مثل الولايات المتحدة وسويسرا نظام إضافة الفلوريد إلى مياه الشرب. وتصرف الولايات المتحدة في هذا السبيل نحو 0.72 دولار لكل شخص في السنة (حسب أرقام عام 1999). وتنصح هيئة الصحة العالمية إضافة الفلوريد إلى الماء في حيز بين 5و0 - 1.0 مليجرام لكل لتر ماء.
وبالنسبة للمياه المعدنية التي تعبأ في زجاجات فهي لا تفصح في العادة عن كمية الفلوريد في الماء. كما أن الماء المحضر بطريقة تحلية المياه يخلو من أملاح الفلوريد حيث تنفصل عن الماء خلال عملية تخليصه من الأملاح.
الهدف
التنفيذ

Fluoridation does not affect the appearance, taste, or smell of drinking water.[1] It is normally accomplished by adding one of three compounds to the water: sodium fluoride, fluorosilicic acid, or sodium fluorosilicate.
- Sodium fluoride (NaF) was the first compound used and is the reference standard.[2] It is a white, odorless powder or crystal; the crystalline form is preferred if manual handling is used, as it minimizes dust.[3] It is more expensive than the other compounds, but is easily handled and is usually used by smaller utility companies.[4]
- Fluorosilicic acid (H2SiF6) is an inexpensive liquid by-product of phosphate fertilizer manufacture.[2] It comes in varying strengths, typically 23–25%; because it contains so much water, shipping can be expensive.[3] It is also known as hexafluorosilicic, hexafluosilicic, hydrofluosilicic, and silicofluoric acid.[2]
- Sodium fluorosilicate (Na2SiF6) is a powder or very fine crystal that is easier to ship than fluorosilicic acid. It is also known as sodium silicofluoride.[3]
These compounds were chosen for their solubility, safety, availability, and low cost.[2] A 1992 census found that, for U.S. public water supply systems reporting the type of compound used, 63% of the population received water fluoridated with fluorosilicic acid, 28% with sodium fluorosilicate, and 9% with sodium fluoride.[5] The Centers for Disease Control and Prevention has developed recommendations for water fluoridation that specify requirements for personnel, reporting, training, inspection, monitoring, surveillance, and actions in case of overfeed, along with technical requirements for each major compound used.[6]
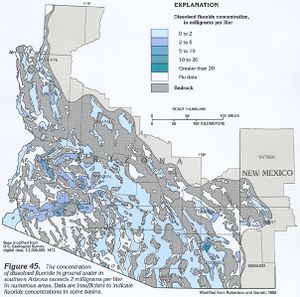
Although fluoride was once considered an essential nutrient, the U.S. National Research Council has since removed this designation due to the lack of studies showing it is essential for human growth, though still considering fluoride a "beneficial element" due to its positive impact on oral health.[7] The U.S. specifies the optimal level of fluoride to range from 0.7 to 1.2 mg/L (milligrams per liter, equivalent to parts per million), depending on the average maximum daily air temperature; the optimal level is lower in warmer climates, where people drink more water, and is higher in cooler climates.[8] The U.S. standard, adopted in 1962, is not appropriate for all parts of the world and is based on assumptions that have become obsolete with the rise of air conditioning and increased use of soft drinks, processed food, and other sources of fluorides. In 1994 a World Health Organization expert committee on fluoride use stated that 1.0 mg/L should be an absolute upper bound, even in cold climates, and that 0.5 mg/L may be an appropriate lower limit.[9] A 2007 Australian systematic review recommended a range from 0.6 to 1.1 mg/L.[10]
الآلية
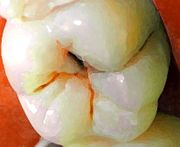
Fluoride exerts its major effect by interfering with the demineralization mechanism of tooth decay. Tooth decay is an infectious disease, the key feature of which is an increase within dental plaque of bacteria such as Streptococcus mutans and Lactobacillus. These produce organic acids when carbohydrates, especially sugar, are eaten.[11] When enough acid is produced so that the pH goes below 5.5,[12] the acid dissolves carbonated hydroxyapatite, the main component of tooth enamel, in a process known as demineralization. After the sugar is gone, some of the mineral loss can be recovered—or remineralized—from ions dissolved in the saliva. Cavities result when the rate of demineralization exceeds the rate of remineralization, typically in a process that requires many months or years.[11]
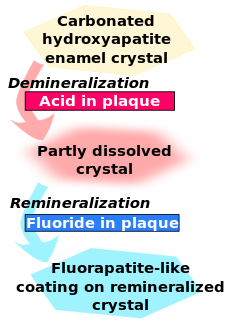
All fluoridation methods, including water fluoridation, create low levels of fluoride ions in saliva and plaque fluid, thus exerting a topical or surface effect. A person living in an area with fluoridated water may experience rises of fluoride concentration in saliva to about 0.04 mg/L several times during a day.[13] Technically, this fluoride does not prevent cavities but rather controls the rate at which they develop.[14] When fluoride ions are present in plaque fluid along with dissolved hydroxyapatite, and the pH is higher than 4.5,[12] a fluorapatite-like remineralized veneer is formed over the remaining surface of the enamel; this veneer is much more acid-resistant than the original hydroxyapatite, and is formed more quickly than ordinary remineralized enamel would be.[11] The cavity-prevention effect of fluoride is mostly due to these surface effects, which occur during and after tooth eruption.[15] Although some systemic (whole-body) fluoride returns to the saliva via blood plasma, and to unerupted teeth via plasma or crypt fluid, there is little data to determine what percentages of fluoride's anticavity effect comes from these systemic mechanisms.[16] Also, although fluoride affects the physiology of dental bacteria,[17] its effect on bacterial growth does not seem to be relevant to cavity prevention.[18]
السلامة
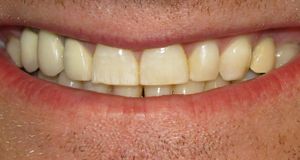
البدائل

- يمكن أيضا إضافة أملاح الفلوريد إلى ملح الطعام أو اللبن أو لمعجون الأسنان.
الاقتصاديات
الأخلاق والسياسة

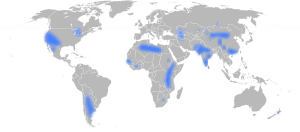
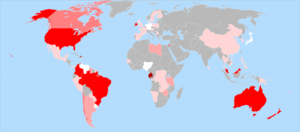
الاستخدام حول العالم
 مقالة مفصلة: الفلورة حسب البلد
مقالة مفصلة: الفلورة حسب البلد
التاريخ

في 25 يناير 1945، أصبحت گراند راپيدز، مشيگن أول مدينة أمريكية تقوم بفلردة مياه الشرب. فبغرض تقليل تسوس الأسنان، أضافت جزء واحد في المليون من الفلوريد إلى الماء.
الهامش
- ^ أ ب Lamberg M, Hausen H, Vartiainen T. Symptoms experienced during periods of actual and supposed water fluoridation. Community Dent Oral Epidemiol. 1997;25(4):291–5. doi:10.1111/j.1600-0528.1997.tb00942.x. PMID 9332806.
- ^ أ ب ت ث Reeves TG. Centers for Disease Control. Water fluoridation: a manual for engineers and technicians [PDF]; 1986 [cited 2008-12-10].
- ^ أ ب ت Lauer WC. Water Fluoridation Principles and Practices. 5th ed. Vol. M4. American Water Works Association; 2004. (Manual of Water Supply Practices). ISBN 1-58321-311-2. History, theory, and chemicals. p. 1–14.
- ^ Nicholson JW, Czarnecka B. Fluoride in dentistry and dental restoratives. In: Tressaud A, Haufe G, editors. Fluorine and Health. Elsevier; 2008. ISBN 978-0-444-53086-8. p. 333–78.
- ^ Division of Oral Health, National Center for Prevention Services, CDC. Fluoridation census 1992 [PDF]. 1993 [cited 2008-12-29].
- ^ Centers for Disease Control and Prevention. Engineering and administrative recommendations for water fluoridation, 1995. MMWR Recomm Rep. 1995;44(RR-13):1–40. PMID 7565542.
- ^ Burt BA (1992). "The changing patterns of systemic fluoride intake". J. Dent. Res. 71 (5): 1228–37. PMID 1607439.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Bailey W, Barker L, Duchon K, Maas W. Populations receiving optimally fluoridated public drinking water—United States, 1992–2006. MMWR Morb Mortal Wkly Rep. 2008;57(27):737–41. PMID 18614991.
- ^ WHO Expert Committee on Oral Health Status and Fluoride Use. Fluorides and oral health [PDF]. 1994.
- ^ أ ب National Health and Medical Research Council (Australia). A systematic review of the efficacy and safety of fluoridation [PDF]. 2007 [cited 2009-10-13]. ISBN 1864964154. Summary: Yeung CA. A systematic review of the efficacy and safety of fluoridation. Evid Based Dent. 2008;9(2):39–43. doi:10.1038/sj.ebd.6400578. PMID 18584000. Lay summary: NHMRC, 2007.
- ^ أ ب ت ث Featherstone JD. Dental caries: a dynamic disease process. Aust Dent J. 2008;53(3):286–91. doi:10.1111/j.1834-7819.2008.00064.x. PMID 18782377.
- ^ أ ب Cury JA, Tenuta LM. How to maintain a cariostatic fluoride concentration in the oral environment. Adv Dent Res. 2008;20(1):13–6. doi:10.1177/154407370802000104. PMID 18694871.
- ^ Pizzo G, Piscopo MR, Pizzo I, Giuliana G. Community water fluoridation and caries prevention: a critical review. Clin Oral Investig. 2007;11(3):189–93. doi:10.1007/s00784-007-0111-6. PMID 17333303.
- ^ Aoba T, Fejerskov O. Dental fluorosis: chemistry and biology. Crit Rev Oral Biol Med. 2002;13(2):155–70. doi:10.1177/154411130201300206. PMID 12097358.
- ^ Hellwig E, Lennon AM. Systemic versus topical fluoride [PDF]. Caries Res. 2004;38(3):258–62. doi:10.1159/000077764. PMID 15153698.
- ^ Tinanoff N. Uses of fluoride. In: Berg JH, Slayton RL, editors. Early Childhood Oral Health. Wiley-Blackwell; 2009. ISBN 978-0-8138-2416-1. p. 92–109.
- ^ Koo H. Strategies to enhance the biological effects of fluoride on dental biofilms. Adv Dent Res. 2008;20(1):17–21. doi:10.1177/154407370802000105. PMID 18694872.
- ^ Marquis RE, Clock SA, Mota-Meira M. Fluoride and organic weak acids as modulators of microbial physiology. FEMS Microbiol Rev. 2003;26(5):493–510. doi:10.1016/S0168-6445(02)00143-2. PMID 12586392.
- ^ خطأ استشهاد: وسم
<ref>غير صحيح؛ لا نص تم توفيره للمراجع المسماةJones-PH - ^ خطأ استشهاد: وسم
<ref>غير صحيح؛ لا نص تم توفيره للمراجع المسماةextent - ^ Douglas WA. History of Dentistry in Colorado, 1859–1959. Denver: Colorado State Dental Assn; 1959. OCLC 5015927. p. 199.