دايدانوسين
 | |
| البيانات السريرية | |
|---|---|
| فئة السلامة أثناء الحمل |
|
| مسارات الدواء | فموي |
| رمز ATC | |
| الحالة القانونية | |
| الحالة القانونية | |
| بيانات الحركية الدوائية | |
| التوافر الحيوي | 30 to 54% |
| ارتباط الپروتين | أقل من 5% |
| Elimination half-life | 1.5 hours |
| الإخراج | كلوي |
| المعرفات | |
| |
| رقم CAS | |
| PubChem CID | |
| DrugBank | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.129.182 |
| Chemical and physical data | |
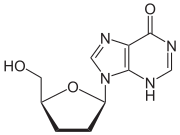
| التركيب | C10H12N4O3 |
| الكتلة المولية | 236.227 g/mol |
دايدانوسين Didanosine (2',3'-dideoxyinosine, ddI) يباع تحت الأسماء التجارية Videx و Videx EC. و هو يعد reverse transcriptase inhibitor, فعال ضدHIV ويستعمل مع تركيب من الأدوية كجزء من antiretroviral drug شديد الفعاليةantiretroviral therapy (HAART).
التاريخ
اكتشف العقار، ورخص بواسطة موريس روبنز، الأستاذ في جامعة بريگم ينگ، عندما كان يحصل على درجته PhD research at Arizona State University in 1988 with David L. J. Tyrrell and Satoru Suzuki. [1] و قد طور بعد ذلك بواسطة Samuel Broder, Hiroaki Mitsuya, و Robert Yarchoan in the National Cancer Institute (NCI) الى المركب الذى يسوق حاليا. حيث لاتستطيعNCI تسويق مركب, و أعطى ترخيص متميز و تسويق لمدة 10 سنوات National Institutes of Health (NIH) Bristol-Myers Squibb Co. (BMS) ddI as Videx tablets.
و أعتبر داييدانوزين ثانى دواء يتم إقراره لعلاج عدوى HIV في بلدان أخرى, بالإضافة الى الولايات المتحدة الأميريكية بواسطة هيئة الأدوية و الأغذية الأميريكية , Food and Drug Administration (FDA) ف 9 أكتوبر , 1991. وقد أسهم هذا الفعل من (FDA) في خفض سعلر zidovudine (AZT), وهو أول دواء مضادanti-HIV .
Didanosine له قدرة ضئيلة غلى مقاومة الأحماض ويتكسر بسهولة بواسطة حمض المعدة damaged by stomach acid. فإن التركيبة الأصلية الت أقرتها FDA تستعمل أقراصا قابلة للمضغ, تحوى داخلها مضاد للحمض antacid , مركب يضاد فعل الحمض المعدى . The chewable tablets were not only large and fragile, they also were foul-tasting and the buffering compound would cause diarrhea. Although the FDA had not approved the original formulation for once-a-day dosing it was possible for some people to take it that way.
At the end of its ten-year license, BMS re-formulated Videx as Videx EC and patented that, which reformulation the FDA approved in 2000. The new formulation is a smaller capsule containing coated microspheres instead of using a buffering compound. It is approved by the FDA for once-a-day dosing. Also at the end of that ten-year period, the NIH licensed didanosine to Barr Laboratories under a non-exclusive license, and didanosine became the first generic anti-HIV drug marketed in the United States.
One of the patents for ddI will expire in the United States on 2006-08-29, but other patents extend beyond that time.
نظرية العمل
Didanosine (ddI) is a nucleoside analogue of adenosine. It differs from other nucleoside analogues, because it does not have any of the regular bases, instead it has hypoxanthine attached to the sugar ring. Within the cell, ddI is phosphorylated to the active metabolite of dideoxyadenosine triphosphate, ddATP, by cellular enzymes. Like other anti-HIV nucleoside analogs, it acts as a chain terminator by incorporation and inhibits viral reverse transcriptase by competing with natural dATP.
الحراك الدوائى
Oral absorption of didanosine is fairly low (42%)[1] but rapid. Food substantially reduces didanosine bioavailability, and the drug should be administered on an empty stomach.[1] The half-life in plasma is only 1.5 hours,[1] but in the intracellular environment more than 12 hours. An enteric-coated formulation is now marketed as well. Elimination is predominantly renal; the kidneys actively secrete didanosine, the amount being 20% of the oral dose.
التفاعل مع الأدوية الأخرى
- A significant interaction has also been recorded with allopurinol, and administration of these drugs together should be avoided.[1]
- Indinavir and delavirdine show reduced in plasma levels when administered simultaneously with didanosine; these drugs should be administered at different times.[1]
- Ketoconazole, itraconazole, ciprofloxacin should be administered at a different time from didanosine due to interactions with the buffering agent.[1]
- Administration with drugs with overlapping toxicity, such as zalcitabine and stavudine, is not recommended.[2]
- Alcohol can exacerbate didanosine's toxicity, and avoiding drinking alcohol while taking didanosine is recommended.[1]
آثار جانبية
The most common adverse events with didanosine are diarrhea, nausea, vomiting, abdominal pain, fever, headache and rash. Peripheral neuropathy occurred in 21-26% of participants in key didanosine trials.[1]
Pancreatitis is rarely observed but has caused occasional fatalities, and has black box warning status. Other reported serious adverse events are retinal changes, optic neuritis and alterations of liver functions. The risk of some of these serious adverse events is increased by drinking alcohol.
المقاومة
Drug resistance to didanosine does develop, though slower than to zidovudine (AZT). The most common mutation observed in vivo is L74V in the viral pol gene, which confers cross-resistance to zalcitabine; other mutations observed include K65R and M184V .[1][3]
المصادر
- ^ أ ب ت ث ج ح خ د ذ VIDEX (didanosine): chewable/dispersible buffered tablets; buffered powder for oral solution; pediatric powder for oral solution. Product information (July 2000)
- ^ DHHS panel. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents (May 4, 2006). (Available for download from AIDSInfo)
- ^ Moyle GJ. Use of viral resistance patterns to antiretroviral drugs in optimising selection of drug combinations and sequences. Drugs 1996;52:168-185
قراءات إضافية
- Yarchoan R, Mitsuya H, Broder S. AIDS therapies. Sci Am 1988;259(4):110-9
- Männistö P.T., Tuominen R.K. in Farmakologia ja Toksikologia, 5th edition: (ed. Koulu, Tuomisto, Paasonen) Medicina, 1996
- Rang H.P., Dale M.M., Ritter J.M.: Pharmacology, 3rd edition. Pearson Professional Ltd, 1995
- Watson et al.: Molecular Biology of the Gene 4th edition. The Benjamin/Cummings Publishing Company, 1987
- Mitsuya H, Yarchoan R, Broder S. Molecular targets for AIDS therapy. Science 1990;249(4976):1533-44
- Yarchoan R, Mitsuya H, Thomas RV, et al. In vivo activity against HIV and favorable toxicity profile of 2',3'-dideoxyinosine. Science 1989;245(4916):412-5
- NIH Oral History of Samuel Broder describing development of AIDS drugs
- NIH Oral History of Robert Yarchoan describing development of AIDS drugs
- NIH Office of Technology Transfer Report on Development and Licensing of ddI
- FDA reviews safety of two HIV drugs
- ECHA InfoCard ID from Wikidata
- Infobox-drug molecular-weight unexpected-character
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
- مثبطات ترانسكريپتاز العكسي المضاهية للنيكليوسيد
- پيورينات
- بريستول-مايرز سكويب
- الأدوية الأساسية حسب منظمة الصحة العالمية