إنتگرين
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
إنتگرين integrin أو مستقبلات الإنتگرين integrin receptor ، هي عبارة بروتينات غشائية مدمجة في الغشاء الپلازمي للخلايا الحية. يلعب الإنتگرين دورا أساسيا في ارتباط الخلايا ببعضها وبالموجد خارج الخلية extracellular matrix (خاصة في الدليل المحوري لمخروط النمو) وأيضا في التحاس signal transduction من الموجد خارج الخلية إلى الخلية الحية. توجد أنواع مختلفة من الانتگرينات والعديد من الخلايا تملك أنماطا مختلفة منها على سطوحها الخلوية، حيث تعد ذات دور حيوي أساسي بالنسبة لمعظم الاحياء من الإسفنجيات إلى الإنسان.
تلعب الإنتجرينات، جنباً مع جنب مع بروتينات أخرى مثل الكادهيرينات وجزيئات التصاق الخلية والسلكتينات للتوسط في تفاعلات واتصال خلية-خلية/خلية/موجد. وتربط الإنتجرينات سطح الخلية بمكونات الموجد خارج الخلية ECM مثل فيبرونكتين وڤيترونكتين وكولاجين ولامينين.
البنية
Integrins are obligate heterodimers composed of α and β subunits. Several genes code for multiple isoforms of these subunits, which gives rise to an array of unique integrins with varied activity. In mammals, integrins are assembled from eighteen α and eight β subunits,[3] in Drosophila five α and two β subunits, and in Caenorhabditis nematodes two α subunits and one β subunit.[4] The α and β subunits are both class I transmembrane proteins, so each penetrates the plasma membrane once, and can possess several cytoplasmic domains.[5]
| الجين | البروتين | المترادفات |
|---|---|---|
| ITGA1 | CD49a | VLA1 |
| ITGA2 | CD49b | VLA2 |
| ITGA3 | CD49c | VLA3 |
| ITGA4 | CD49d | VLA4 |
| ITGA5 | CD49e | VLA5 |
| ITGA6 | CD49f | VLA6 |
| ITGA7 | ITGA7 | FLJ25220 |
| ITGA8 | ITGA8 | |
| ITGA9 | ITGA9 | RLC |
| ITGA10 | ITGA10 | PRO827 |
| ITGA11 | ITGA11 | HsT18964 |
| ITGAD | CD11D | FLJ39841 |
| ITGAE | CD103 | HUMINAE |
| ITGAL | CD11a | LFA1A |
| ITGAM | CD11b | MAC-1 |
| ITGAV | CD51 | VNRA, MSK8 |
| ITGA2B | CD41 | GPIIb |
| ITGAX | CD11c |
| الجين | البروتين | المترادفات |
|---|---|---|
| ITGB1 | CD29 | FNRB, MSK12, MDF2 |
| ITGB2 | CD18 | LFA-1, MAC-1, MFI7 |
| ITGB3 | CD61 | GP3A, GPIIIa |
| ITGB4 | CD104 | |
| ITGB5 | ITGB5 | FLJ26658 |
| ITGB6 | ITGB6 | |
| ITGB7 | ITGB7 | |
| ITGB8 | ITGB8 |
Variants of some subunits are formed by differential RNA splicing; for example, four variants of the beta-1 subunit exist. Through different combinations of the α and β subunits, 24 unique mammalian integrins are generated, excluding splice- and glycosylation variants.[6]
Integrin subunits span the cell membrane and have short cytoplasmic domains of 40–70 amino acids. The exception is the beta-4 subunit, which has a cytoplasmic domain of 1,088 amino acids, one of the largest of any membrane protein. Outside the cell membrane, the α and β chains lie close together along a length of about 23 nm; the final 5 nm N-termini of each chain forms a ligand-binding region for the ECM. They have been compared to lobster claws, although they don't actually "pinch" their ligand, they chemically interact with it at the insides of the "tips" of their "pinchers".
The molecular mass of the integrin subunits can vary from 90 kDa to 160 kDa. Beta subunits have four cysteine-rich repeated sequences. Both α and β subunits bind several divalent cations. The role of divalent cations in the α subunit is unknown, but may stabilize the folds of the protein. The cations in the β subunits are more interesting: they are directly involved in coordinating at least some of the ligands that integrins bind.
Integrins can be categorized in multiple ways. For example, some α chains have an additional structural element (or "domain") inserted toward the N-terminal, the alpha-A domain (so called because it has a similar structure to the A-domains found in the protein von Willebrand factor; it is also termed the α-I domain). Integrins carrying this domain either bind to collagens (e.g. integrins α1 β1, and α2 β1), or act as cell-cell adhesion molecules (integrins of the β2 family). This α-I domain is the binding site for ligands of such integrins. Those integrins that don't carry this inserted domain also have an A-domain in their ligand binding site, but this A-domain is found on the β subunit.
In both cases, the A-domains carry up to three divalent cation binding sites. One is permanently occupied in physiological concentrations of divalent cations, and carries either a calcium or magnesium ion, the principal divalent cations in blood at median concentrations of 1.4 mM (calcium) and 0.8 mM (magnesium). The other two sites become occupied by cations when ligands bind—at least for those ligands involving an acidic amino acid in their interaction sites. An acidic amino acid features in the integrin-interaction site of many ECM proteins, for example as part of the amino acid sequence Arginine-Glycine-Aspartic acid ("RGD" in the one-letter amino acid code).
Structure
Despite many years of effort, discovering the high-resolution structure of integrins proved to be challenging, as membrane proteins are classically difficult to purify, and as integrins are large, complex and highly glycosylated with many sugar 'trees' attached to them. Low-resolution images of detergent extracts of intact integrin GPIIbIIIa, obtained using electron microscopy, and even data from indirect techniques that investigate the solution properties of integrins using ultracentrifugation and light scattering, were combined with fragmentary high-resolution crystallographic or NMR data from single or paired domains of single integrin chains, and molecular models postulated for the rest of the chains.
The X-ray crystal structure obtained for the complete extracellular region of one integrin, αvβ3,[1] shows the molecule to be folded into an inverted V-shape that potentially brings the ligand-binding sites close to the cell membrane. Perhaps more importantly, the crystal structure was also obtained for the same integrin bound to a small ligand containing the RGD-sequence, the drug cilengitide.[7] As detailed above, this finally revealed why divalent cations (in the A-domains) are critical for RGD-ligand binding to integrins. The interaction of such sequences with integrins is believed to be a primary switch by which ECM exerts its effects on cell behaviour.
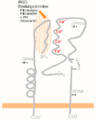
The structure poses many questions, especially regarding ligand binding and signal transduction. The ligand binding site is directed towards the C-terminal of the integrin, the region where the molecule emerges from the cell membrane. If it emerges orthogonally from the membrane, the ligand binding site would apparently be obstructed, especially as integrin ligands are typically massive and well cross-linked components of the ECM. In fact, little is known about the angle that membrane proteins subtend to the plane of the membrane; this is a problem difficult to address with available technologies. The default assumption is that they emerge rather like little lollipops, but there is little evidence for this. The integrin structure has drawn attention to this problem, which may have general implications for how membrane proteins work. It appears that the integrin transmembrane helices are tilted (see "Activation" below), which hints that the extracellular chains may also not be orthogonal with respect to the membrane surface.
Although the crystal structure changed surprisingly little after binding to cilengitide, the current hypothesis is that integrin function involves changes in shape to move the ligand-binding site into a more accessible position, away from the cell surface, and this shape change also triggers intracellular signaling. There is a wide body of cell-biological and biochemical literature that supports this view. Perhaps the most convincing evidence involves the use of antibodies that only recognize integrins when they have bound to their ligands, or are activated. As the "footprint" that an antibody makes on its binding target is roughly a circle about 3 nm in diameter, the resolution of this technique is low. Nevertheless, these so-called LIBS (Ligand-Induced-Binding-Sites) antibodies unequivocally show that dramatic changes in integrin shape routinely occur. However, how the changes detected with antibodies look on the structure is still unknown.
Activation
When released into the cell membrane, newly synthesized integrin dimers are speculated to be found in the same "bent" conformation revealed by the structural studies described above. One school of thought claims that this bent form prevents them from interacting with their ligands, although bent forms can predominate in high-resolution EM structures of integrin bound to an ECM ligand. Therefore, at least in biochemical experiments, integrin dimers must apparently not be 'unbent' in order to prime them and allow their binding to the ECM. In cells, the priming is accomplished by a protein talin, which binds to the β tail of the integrin dimer and changes its conformation.[8][9] The α and β integrin chains are both class-I transmembrane proteins: they pass the plasma membrane as single transmembrane alpha-helices. Unfortunately, the helices are too long, and recent studies suggest that, for integrin gpIIbIIIa, they are tilted with respect both to one another and to the plane of the membrane. Talin binding alters the angle of tilt of the β3 chain transmembrane helix in model systems and this may reflect a stage in the process of inside-out signalling which primes integrins.[10] Moreover, talin proteins are able to dimerize[11] and thus are thought to intervene in the clustering of integrin dimers which leads to the formation of a focal adhesion. Recently, the Kindlin-1 and Kindlin-2 proteins have also been found to interact with integrin and activate it.[12]
الوظيفة
نقل الإشارة
الانتگرين يلعب دورا مهما في التأشير الخلوي cell signaling. فالاتصال بين جزيئات (الموجد خارج الخلية) يمكن ان يحدث إشارة يمكن أن تنقل إلى الخلية عبر كينازات بروتينية متصلة بالنهاية الداخل-خلوية لجزيئات الإنتگرين.
الإشارات التي تستقبلها الخلايا عن طريق الإنتگرين تلعب دورا في :
- نمو الخلايا cell growth
- انقسام الخلايا cell division
- بقيا الخلايا cell survival
- التمايز الخلوي cellular differentiation
- موت الخلية المبرمج (programmed cell death.
إنتگرينات الحيوانات الفقارية
القائمة التالية هي للإنتگرينات التي تم إيجادها في الفقاريات:
| الاسم | مترادفات | التوزيع | لجينات |
| α1β1 | العديد | كولاجينات, لامينينات.[13] | |
| α2β1 | العديد | كولاجينات, لامينينات[13] | |
| α4β1 | VLA-4[13] | Hematopoietic cells | فيبرونيكتين, VCAM-1[13] |
| α5β1 | مستقبل فيبرونيكتين | متوزعة بكثرة | فيبرونكتين[13] وپروتينازات |
| α6β1 | مستقبل لامينين | متوزعة بكثرة | matrix macromolecules لامينينات |
| αLβ2 | LFA-1[13] | T-lymphocytes | ICAM-1, ICAM-2[13] |
| αMβ2 | Mac-1, CR3[13] | خلية وحيدة | بروتينات المصل, ICAM-1[13] |
| αIIbβ3 | صفيحات دموية[13] | فيبرينوجين, فيبرونيكتين[13] | |
| αVβ3 | مستقبل ڤيترونكتين[14] | activated endothelial cells, melanoma, glioblastoma | vitronectin [14], fibronectin, fibrinogen, osteopontin, Cyr61 |
| αVβ5 | متوزعة بكثرة, خصوصاً في الأرومة الليفية, الخلايا الظهارية | ڤيترونكتين and adenovirus | |
| αVβ6 | سرطانات الكبد والرئة | فيبرونيكتين; TGFβ1+3 | |
| α6β4 | خلايا ظهارية[13] | لامينين[13] |
إنتگرينات بيتا1 ترتبط بالعديد من سلاسل الإنتگرين ألفا.
صور اضافية
-
Integrin (captions in German)
الهامش
- ^ أ ب Xiong JP, Stehle T, Diefenbach B, Zhang R, Dunker R, Scott DL, Joachimiak A, Goodman SL, Arnaout MA (October 2001). "Crystal structure of the extracellular segment of integrin alpha Vbeta3". Science. 294 (5541): 339–45. Bibcode:2001Sci...294..339X. doi:10.1126/science.1064535. PMC 2885948. PMID 11546839.
- ^ Sauer FG, Fütterer K, Pinkner JS, Dodson KW, Hultgren SJ, Waksman G (August 1999). "Structural basis of chaperone function and pilus biogenesis". Science. 285 (5430): 1058–61. doi:10.1126/science.285.5430.1058. PMID 10446050.
- ^ Bruce A, Johnson A, Lewis J, Raff M, Roberts K, Walter P (2002). "Integrins". Molecular Biology of the Cell (4th ed.). New York: Garland Science.
- ^ Humphries MJ (2000). "Integrin structure". Biochemical Society Transactions. 28 (4): 311–39. doi:10.1042/0300-5127:0280311. PMID 10961914.
- ^ Nermut MV, Green NM, Eason P, Yamada SS, Yamada KM (December 1988). "Electron microscopy and structural model of human fibronectin receptor". The EMBO Journal. 7 (13): 4093–9. doi:10.1002/j.1460-2075.1988.tb03303.x. PMC 455118. PMID 2977331.
- ^ Hynes RO (September 2002). "Integrins: bidirectional, allosteric signaling machines". Cell. 110 (6): 673–87. doi:10.1016/S0092-8674(02)00971-6. PMID 12297042. S2CID 30326350.
- ^ Smith JW (June 2003). "Cilengitide Merck". Current Opinion in Investigational Drugs. 4 (6): 741–5. PMID 12901235.
- ^ Calderwood DA (June 2004). "Talin controls integrin activation". Biochemical Society Transactions. 32 (Pt3): 434–7. doi:10.1042/BST0320434. PMID 15157154.
- ^ Calderwood DA, Zent R, Grant R, Rees DJ, Hynes RO, Ginsberg MH (October 1999). "The Talin head domain binds to integrin beta subunit cytoplasmic tails and regulates integrin activation". The Journal of Biological Chemistry. 274 (40): 28071–4. doi:10.1074/jbc.274.40.28071. PMID 10497155.
- ^ Shattil SJ, Kim C, Ginsberg MH (April 2010). "The final steps of integrin activation: the end game". Nature Reviews. Molecular Cell Biology. 11 (4): 288–300. doi:10.1038/nrm2871. PMC 3929966. PMID 20308986.
- ^ Goldmann WH, Bremer A, Häner M, Aebi U, Isenberg G (1994). "Native talin is a dumbbell-shaped homodimer when it interacts with actin". Journal of Structural Biology. 112 (1): 3–10. doi:10.1006/jsbi.1994.1002. PMID 8031639.
- ^ Harburger DS, Bouaouina M, Calderwood DA (April 2009). "Kindlin-1 and -2 directly bind the C-terminal region of beta integrin cytoplasmic tails and exert integrin-specific activation effects". The Journal of Biological Chemistry. 284 (17): 11485–97. doi:10.1074/jbc.M809233200. PMC 2670154. PMID 19240021.
- ^ أ ب ت ث ج ح خ د ذ ر ز س ش Molecular cell biology. Lodish, Harvey F. 5. ed. : - New York : W. H. Freeman and Co., 2003, 973 s. b ill. ISBN 0-7167-4366-3
- ^ أ ب PMID: 10037797
وصلات خارجية
 وسائط متعلقة بـIntegrins من مشاع المعرفة.
وسائط متعلقة بـIntegrins من مشاع المعرفة.- The Integrin Protein
- Talin substrate for calpain – PMAP The Proteolysis Map animation.
- MeSH Integrins